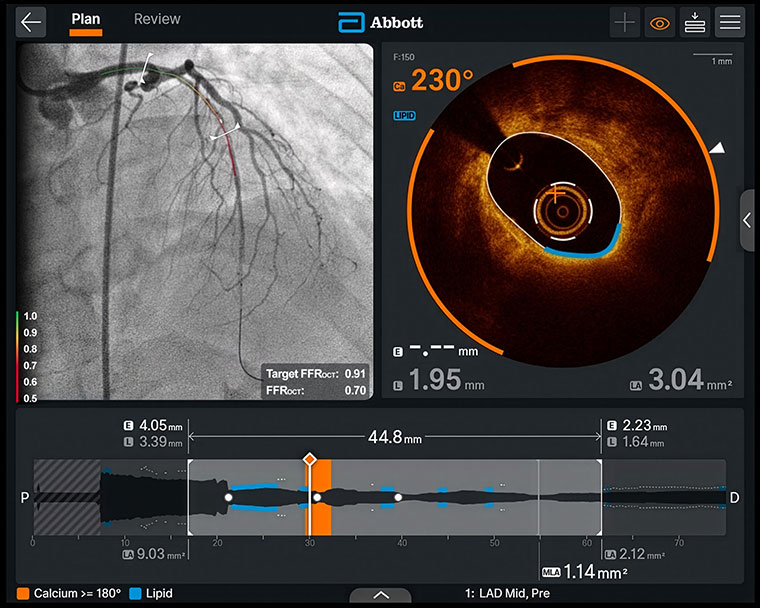
Abbott Laboratories (NYSE: ABT) (ETR: ABL) has made significant progress by receiving both FDA clearance and CE Mark approval for its Ultreon 3.0 AI-powered coronary imaging platform. The medtech company revealed the achievement on Apr. 28.
Based in Illinois, Abbott developed this next-generation software to help doctors during procedures that fix blocked heart arteries without major surgery.
The approvals allow the platform’s use in the United States and Europe. Physicians can adopt the technology to improve decision-making for patients with complex coronary artery disease by combining high-resolution imaging with AI.
Ultreon 3.0 uses what’s called optical coherence tomography (OCT), a technique that employs infrared light to produce detailed cross-sectional images of the inside of coronary arteries. Doctors insert a catheter to capture these images quickly in about one second. The AI software then analyzes the images to identify plaque type, assess blockage size and location, and recommend the best spot and size for a small metal mesh tube (called a stent) that holds the artery open.
It also enables post-procedure checks to confirm improved blood flow. This system gives much clearer pictures than the standard ultrasound method. Notably, it needs very little or even no special dye. Doctors usually inject this dye so they can see the arteries on the X-ray camera. Using less dye (or none) is safer for patients with kidney problems.
“Abbott’s Ultreon 3.0 offers a single, AI-driven platform that quickly and easily captures the images physicians need to fully understand the size and angle of the blockage, and create an actionable plan to help the patient,” commented German cardiologist David M. Leistner in a news release from the medical tech developer.
Read more: Breath Diagnostics advances pre-op pneumonia screening with FDA breakthrough designation
Abbott expands into new diagnostic areas
Beyond cardiovascular technologies, Abbott continues to broaden its portfolio. In March, the company completed its acquisition of Exact Sciences, a move that established a strong position in oncology diagnostics.
This integration has added cancer screening capabilities, particularly through Exact Sciences’ Cancerguard multi-cancer early detection test. This screening tool uses a blood draw to detect signals from over 50 cancer types and subtypes, including several aggressive ones like lung cancer.
The takeover is enabling Abbott to stand alongside other innovators in lung cancer screening and broader multi-cancer detection. Companies such as Breath Diagnostics, creator of the OneBreath breath-based test for early lung cancer; Grail Inc (NASDAQ: GRAL) (FRA: NL0), developer of the ‘Galleri’ blood test for over 50 cancers; and Guardant Health Inc (NASDAQ: GH) (FRA: 5GH), operate in this space.
Abbott now participates in efforts to identify cancers earlier when intervention may improve outcomes.
Abbott reports favourable numbers
Amid this expansion, Abbott reported solid first-quarter 2026 results. Total sales reached US$11.16 billion, a 7.8 per cent year-over-year increase. Medical device sales in particular grew strongly with an 8.5 per cent rise compared to the first quarter of 2025. Earnings per share clocked in at US$1.15.
Following the Exact Sciences acquisition, Abbott updated its full-year 2026 guidance. It projects comparable sales growth of 6.5 per cent to 7.5 per cent and earnings per share ranging from US$5.38 to US$5.58.
Abbott maintains a diversified position across diagnostics, medical devices, nutrition and pharmaceuticals, serving markets in more than 160 countries. Performance has remained steady despite the nutrition segment facing headwinds in the quarter. Nutrition sales declined by 6 per cent year-over-year during the three-month period.
Read more: Prestigious medtech intelligence firm recognizes Breath Diagnostics for innovation
Follow Rowan Dunne on LinkedIn
rowan@mugglehead.com