Shionogi & Co. Ltd (TYO: 4507) has agreed to acquire Apnimed’s remaining stake in their joint sleep-disorder venture, consolidating full control as the partners advance new treatments for obstructive sleep apnea.
Apnimed announced Thursday that it will sell its 50 per cent ownership in Shionogi-Apnimed Sleep Science, LLC (SASS) for upfront and milestone payments. The deal marks a strategic shift as Apnimed narrows its focus to its lead drug candidate, AD109.
Under the agreement, Apnimed will receive USD$100 million in upfront cash. Additionally, it could earn another USD$50 million tied to a development milestone for SASS-002, also known as sulthiame. The company will also collect tiered royalties from any future commercial products derived from SASS intellectual property.
The companies expect to close the transaction in the second quarter of 2026.
Apnimed’s CEO, Larry Miller, said the move allows the company to strengthen its balance sheet. He also indicated the company will prioritize advancing AD109 toward regulatory submission. Furthermore, he described the partnership with Shionogi as productive and expressed confidence in continued progress under sole ownership.
SASS began in 2023 as a joint venture between the two companies. It aimed to develop new treatments for sleep disorders using combined expertise. Shionogi contributed drug discovery capabilities, while Apnimed focused on translational research.
Now, Shionogi will take full ownership and control of the venture’s pipeline.
Meanwhile, Apnimed will direct more resources toward AD109, its primary drug candidate for obstructive sleep apnea (OSA). The company expects to submit a New Drug Application to the U.S. Food and Drug Administration in the second quarter of 2026.
Read more: Luxury hotel in Singapore starts using ‘DeepSleep’ sound tech from NuCalm
Read more: Five-star Singapore hotel integrates sleep technology into its 156 rooms
OSA is a widespread and under-treated condition
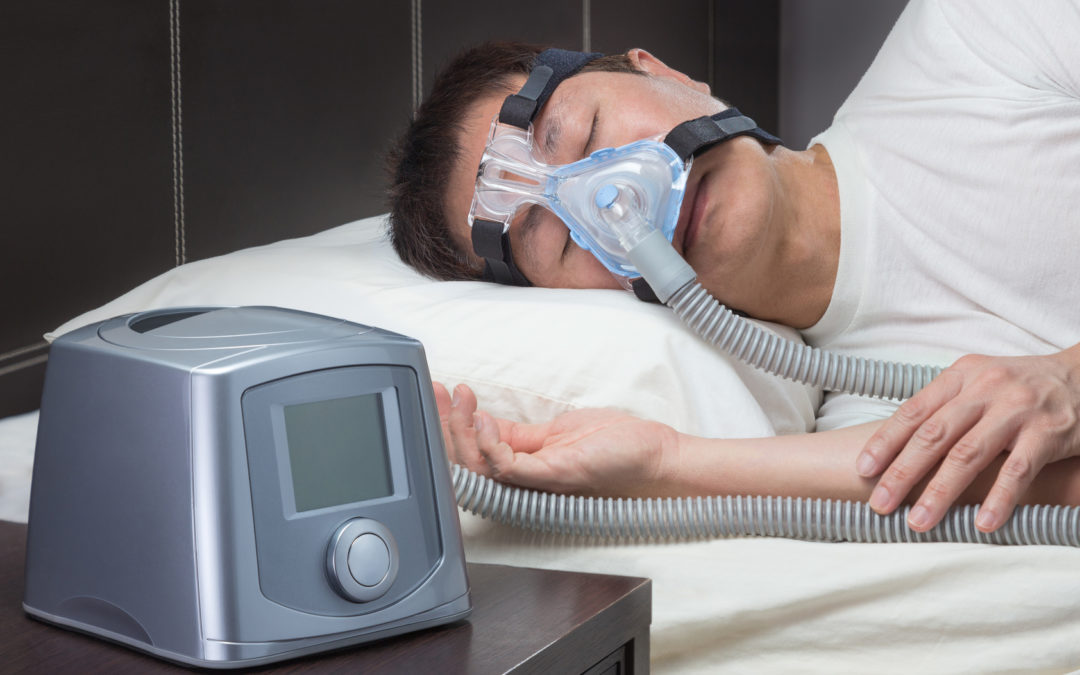
AD109 targets a core biological cause of OSA rather than managing symptoms alone. It combines aroxybutynin, an antimuscarinic compound, with atomoxetine, a norepinephrine reuptake inhibitor. Together, these drugs aim to improve muscle tone in the airway during sleep.
Additionally, the therapy is designed as a once-daily pill taken at bedtime. This approach could reduce reliance on devices like CPAP machines, which many patients find difficult to use consistently.
OSA remains a widespread and under-treated condition. It occurs when the upper airway collapses repeatedly during sleep, reducing oxygen levels in the blood. Consequently, patients often experience disrupted sleep and long-term health risks.
Experts estimate that about 80 million people in the United States live with OSA. Globally, the number reaches roughly one billion. However, many cases remain undiagnosed or untreated.
Furthermore, untreated OSA can lead to serious complications. These include cardiovascular disease, cognitive impairment, and metabolic disorders. In severe cases, it can also increase mortality risk.
Patients with OSA may experience hundreds of breathing interruptions each night. Each event briefly lowers oxygen levels and strains the body. Over time, this repeated stress affects multiple organ systems.
Meanwhile, current treatments often involve mechanical devices or surgical interventions. These options can be effective, but they also present barriers to long-term adherence.
Apnimed aims to offer a simpler alternative through pharmacological treatment. Additionally, AD109’s design could make therapy more accessible to a broader patient population.
Shionogi, for its part, will continue advancing SASS-related programs independently. The company has experience developing small-molecule drugs and bringing them to market. Consequently, it may accelerate progress on candidates like sulthiame.
Read more: Jet lag aid sleep tech app creator ‘Timeshifter’ sets up Denmark hub
Read more: South Korea’s Fine Sleeping Expo celebrates World Sleep Day
Apnimed gains funding and flexibility
The transaction reflects a broader trend in biotech partnerships. Companies often restructure collaborations to sharpen focus or unlock capital. In this case, Apnimed gains funding and flexibility, while Shionogi gains full control of a specialized research platform.
Furthermore, the royalty structure allows Apnimed to retain long-term upside from future products. This approach balances immediate funding needs with potential future returns.
As both companies move forward, the focus remains on addressing unmet needs in sleep medicine. Additionally, the success of AD109 could reshape how clinicians treat obstructive sleep apnea.
.