MiniMed Group Inc (NASDAQ: MMED), formerly Medtronic PLC‘s (NYSE: MDT) (FRA: 2M6) diabetes business unit, began trading as an independent public company on the Nasdaq Global Select Market under the ticker symbol MMED on Mar. 6. This market is the Nasdaq’s premium or top tier.
The initial public offering involved the sale of 28 million shares priced at US$20 each, raising approximately US$560 million before fees with net proceeds of about US$538 million. Trading opened at US$19.05 per share, resulting in a market capitalization of roughly US$5.3 billion on the medtech company’s debut day. The IPO closed on Mar. 9.
This pricing was well below the previously marketed range of US$25 to US$28 (a roughly 20 to 29 per cent discount), reflecting cautious investor demand amid heightened market volatility at the time of the offering.
Medtronic currently retains approximately 90 per cent of MiniMed’s outstanding shares following the offering and has indicated plans for a full separation at a later date, potentially through a split-off transaction. This type of deal would see that Medtronic fully separates from the company at some point in the future by offering its own shareholders the choice to exchange some or all of their Medtronic shares for MiniMed shares.
MiniMed develops and sells insulin delivery systems and continuous glucose monitoring devices for people with diabetes. The company operates in more than 80 countries and reported revenue of US$2.89 billion in its most recent fiscal year.
“What hasn’t changed is why we’re here,” said MiniMed CEO Que Dallara. “For more than 40 years, our mission has been to make every day a better day for people with diabetes.”
“For more than 40 years, we have been pushing the envelope on outcomes and burden reduction.”@MiniMed CEO Que Dallara shares the $MMED journey to listing day. pic.twitter.com/NleicJYjno
— Nasdaq Exchange (@NasdaqExchange) March 10, 2026
Read more: Breath Diagnostics advances pre-op pneumonia screening with FDA breakthrough designation
News builds on regulatory approvals in EU and U.S.
MiniMed’s product portfolio includes automated insulin delivery systems that use sensors to monitor glucose levels and insulin pumps to adjust dosing.
In Europe, the company recently received CE Mark approval for its MiniMed 780G system integrated with the Instinct continuous glucose sensor from Abbott Laboratories (NYSE: ABT) (ETR: ABL). The Instinct sensor is designed to last up to 15 days and is indicated for use in children as young as 2 years old.
CE Mark approval validates that a product is in compliance with EU safety, health and environmental protection standards. The company has stated that the integrated system is expected to become available in European markets this summer.
In the United States, the Food and Drug Administration also granted approval in September of 2025 for this system, including an expanded indication for type 2 diabetes.
More recently, the FDA gave its assent to the MiniMed Go, a device intended for manual insulin dosing that pairs the Instinct sensor with a connected smart insulin pen. MiniMed is planning to launch this gadget in the American market during the spring months of 2026.
These approvals expand the company’s available options in both the European and U.S. markets as it begins operating independently. They also position MiniMed to compete more aggressively against specialized rivals such as DexCom Inc (NASDAQ: DXCM) (FRA: DC4), Abbott, Insulet Corp (NASDAQ: PODD) (FRA: GOV) and Tandem Diabetes Care Inc (NASDAQ: TNDM) (FRA: TD5A) in the automated insulin delivery and glucose monitoring markets.
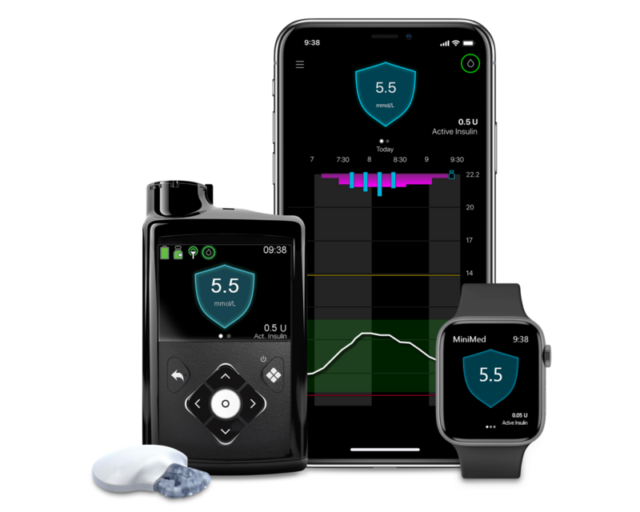
MiniMed 780G system. Image credit: Medtronic
Read more: Prestigious medtech intelligence firm recognizes Breath Diagnostics for innovation
Follow Rowan Dunne on LinkedIn
rowan@mugglehead.com














