In just four years, the landscape of psychedelic drug collaborations has undergone a remarkable transformation, with over 100 drug development partnerships emerging since 2019.
This surge, representing a staggering 500 per cent increase in 2023 alone, is intricately linked to a shifting regulatory environment that increasingly acknowledges the clinical potential of illicit substances such as psilocybin, LSD and ketamine.
A recent report by the market analysis firm GlobalData showed the relationship between regulatory advancements and the burgeoning alliances within the psychedelic drug industry.
The popularity of psychedelics keeps on rising as the substances show positive results in clinical trials. The drugs continue to demonstrate their potential to treat certain mental health conditions such as depression and anxiety as well as other metabolic disorders.
“Which could accelerate innovative psychedelic drug development in the future,” GlobalData analyst Alison Labya said.
The United States had 63 partnerships, while Canada saw 29 and the United Kingdom followed with 15 partnerships.
In March 2023, Senators Cory Booker and Rand Paul introduced a bipartisan Breakthrough Therapies Act to amend the Controlled Substances Act, potentially reclassifying therapeutic psychedelics.
Financial support for psychedelic research is evident in a $70 million deal between Vine Ventures a venture capital firm and the prominent advocacy organization MAPS, indicating a trend toward nonprofit and commercial partnerships.
Psychedelics-related startups secured over $236 million between July 2021 and July 2022, with a focus on advancing therapeutic options through the Food and Drug Administration’s clinical trial pipeline.
In August 2020, Health Canada was the first regulatory agency to approve a clinical trial for psilocybin-assisted therapy under Canada’s Special Access Program.
But it wasn’t until this year that the first country legalized the substances for medical use. End of June, Australia passed a bill that approved psychiatrists to prescribe MDMA or psilocybin for mental health disorders.
The Oceanian country has also seen growth in the psychedelics industry such as the private company Psylo signing a development partnership with Daiichi Sankyo to advance its non-hallucinogenic psychedelic drugs indicated for chronic mental illnesses.
“The increase of partnership deals involving innovator psychedelic drugs over the last five years could see further clinical trials with the potential to gain regulatory approval in indications such as depression, ADHD, eating disorders and addiction,” said Labya.
“However, biopharmaceutical companies developing psychedelic therapeutics require careful consideration of psychedelic drug clinical trial design for a favourable risk-to-benefit ratio to enable bringing their drugs to market.”
In Canada, Clearmind Medicine Inc. (CNSX: CMND) entered a co-development partnership with Johns Hopkins University School of Medicine to test an experimental psychedelic for alcohol use disorder. Previously, the company announced a partnership with the Hebrew University of Jerusalem and the Israeli company SciSparc to evaluate an experimental psychedelic and cannabinoid for obesity and metabolic syndrome.
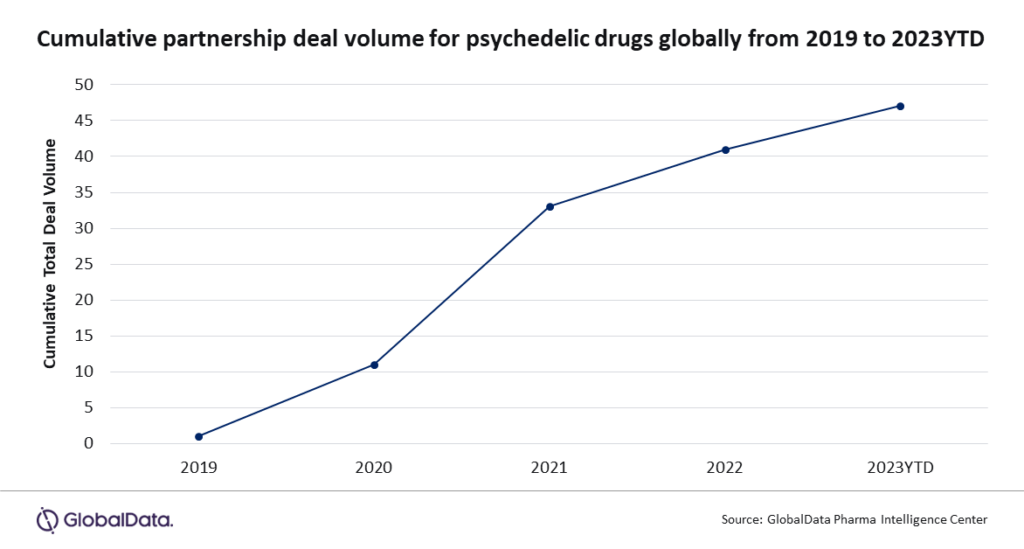
Partnerships of psychedelic drugs growing over time. Chart includes all announced and completed partnership deals from 2019-2023 Graph via GlobalData.
Read more: Stella acquires Field Trip’s U.S. assets to enhance its psychedelic therapy offerings
August was a hot month for psychedelic company acquisitions
In September, Lucy Scientific Discovery Inc. (NASDAQ: LSDI) entered into an all-stock transaction to acquire the wellness company BlueSky Wellness Inc.
Otsuka Pharmaceutical Co. Ltd. (TYO: 4578) announced an investment of US$80 million to acquire Mindset Pharma Inc. (CSE: MSET) (FSE: 9DF) (OTCQB: MSSTF). In the same month, Silo Wellness, Inc. (CSE: SILO) (OTCQB: SILFF) (FSE: 3K7A) also announced its intention to acquire the Jamaica-based psychedelics company NUGL Inc. (OTC Pink: NUGL) (dba Kaya Jamaica) in a transaction valued at $43.3 million.
The Canadian company Cybin Inc. (NYSE American: CYBN) (NEO: CYBN) announced last August it will be acquiring the United Kingdom’s DMT research company Small Pharma Inc. (TSX-V: DMT) (OTCQB: DMTTF), creating an entity with one of the largest intellectual property portfolios in the industry.
As biopharmaceutical companies navigate the complex landscape of psychedelic drug development, the continued rise in popularity highlights a paradigm shift, where once-stigmatized substances emerge as potent tools for addressing some of society’s most pressing mental health challenges.
Natalia@mugglehead.com














