Genprex Inc (NASDAQ: GNPX) (FRA: 2DE) has teamed up with The University of Texas MD Anderson Cancer Center to study two biomarkers in tumours that may predict which lung cancer patients respond best to its gene therapy drug, Reqorsa.
Announced on Tuesday, the partnership will see that researchers examine TROP2 and PTEN proteins to learn why some patients see their cancer shrink or stay stable for a long time while others do not. The goal is to improve patient selection for two ongoing clinical trials so doctors can match the treatment more effectively to people with non-small cell and small cell lung cancers.
Genprex is a small clinical-stage company based in Texas that develops gene therapies for cancer and diabetes. MD Anderson is one of the world’s top cancer hospitals and research centres, located in Houston. It focuses on patient care, prevention and testing new treatments through hundreds of clinical trials.
“It is important to understand why some patients are responding very well to REQORSA,” said Chief Medical Officer Mark Berger, “and we believe that the identification of biomarkers will allow us to predict which future patients may have these similar, positive responses.”
Read more: Breath Diagnostics advances pre-op pneumonia screening with FDA breakthrough designation
Collaboration builds on extensive lung cancer drug research
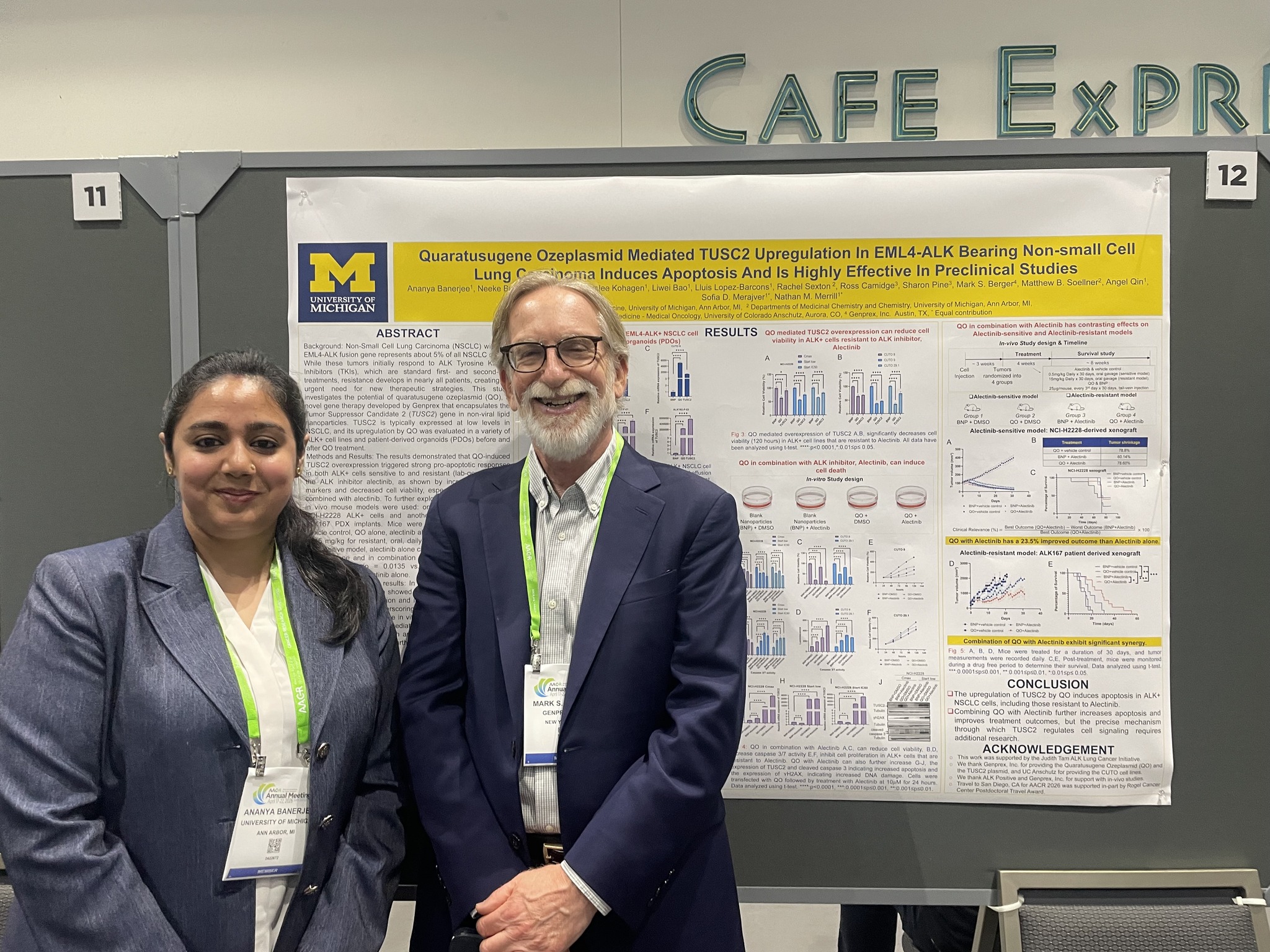
These new studies continue a partnership with MD Anderson that began in 2010. The latest development directly follows positive preclinical data that Genprex’s collaborators presented at the 2026 AACR Annual Meeting in San Diego, California.
Those studies showed that low TROP2 and high PTEN levels can signal resistance to Reqorsa in some lung tumours. When scientists boosted TROP2 in lab tests, the therapy triggered more cancer cell death.
The data also revealed that Reqorsa restores a key tumour suppressor gene called TUSC2, which shrinks tumours, induces cell death and boosts natural killer cells in the immune system. This occurs even in cases resistant to other lung cancer drugs.
The work supports Genprex’s “Acclaim-1” and Acclaim-3 trials. In Acclaim-1, doctors combine Reqorsa with an established targeted drug for patients with advanced non-small cell lung cancer whose disease progressed after prior treatment. Early results showed the therapy was safe, with some patients staying progression-free for months and one holding a partial response for about 42 months.
In Acclaim-3, Reqorsa pairs with an immunotherapy drug as maintenance treatment for extensive-stage small cell lung cancer. One patient achieved a partial remission and continued treatment for more than 18 months. Both trials carry FDA fast-track status.
Read more: Prestigious medtech intelligence firm recognizes Breath Diagnostics for innovation
Genprex stock slides despite progress
The firm remains in the small biotech category with a market value of roughly US$10.5 million. Its stock is currently trading near a 52-week low and shows high volatility.
The company currently reports no revenue, burns through cash at a high rate with ongoing net losses and will likely need to raise more money soon through stock sales. This creates a constant risk of dilution for shareholders. Biotech companies like Genprex also face the ever-present danger that clinical trials could hit delays, produce disappointing results, or fail to meet endpoints, which could quickly erode investor confidence and make future funding even harder to secure.
On the positive side though, the firm maintains solid short-term cash reserves that cover its immediate needs as it advances clinical work.
Genprex’s current optimism stems from recent positive preclinical data on Reqorsa, new biomarker research with MD Anderson, upcoming data presentations at major conferences like ASCO 2026 and strengthened patents in Japan and Europe that protect its combination therapies.
Read more: Breath Diagnostics completes install of advanced mass spectrometry system
Follow Rowan Dunne on LinkedIn
rowan@mugglehead.com