Vitestro, a Netherlands-based medical robotics company, just closed an oversubscribed Series B funding round worth US$70 million.
The company announced the raise on Mar. 10. New strategic investors such as the Labcorp Venture Fund, Mayo Clinic and Sutter Health participated alongside local investment firms PGGM and ROM Utrecht and financial backers like InterVest, MGFO and Puma Venture Capital.
Vitestro plans to use the funds to advance its Aletta autonomous blood-draw device toward U.S. FDA De Novo clearance, expand manufacturing and prepare for commercial launches that will begin in Europe before reaching the United States.
The technology addresses persistent challenges in healthcare, including chronic staffing shortages and inconsistent blood sample quality in what remains the most frequently performed invasive procedure worldwide. Billions of blood draws are completed around the globe per year.
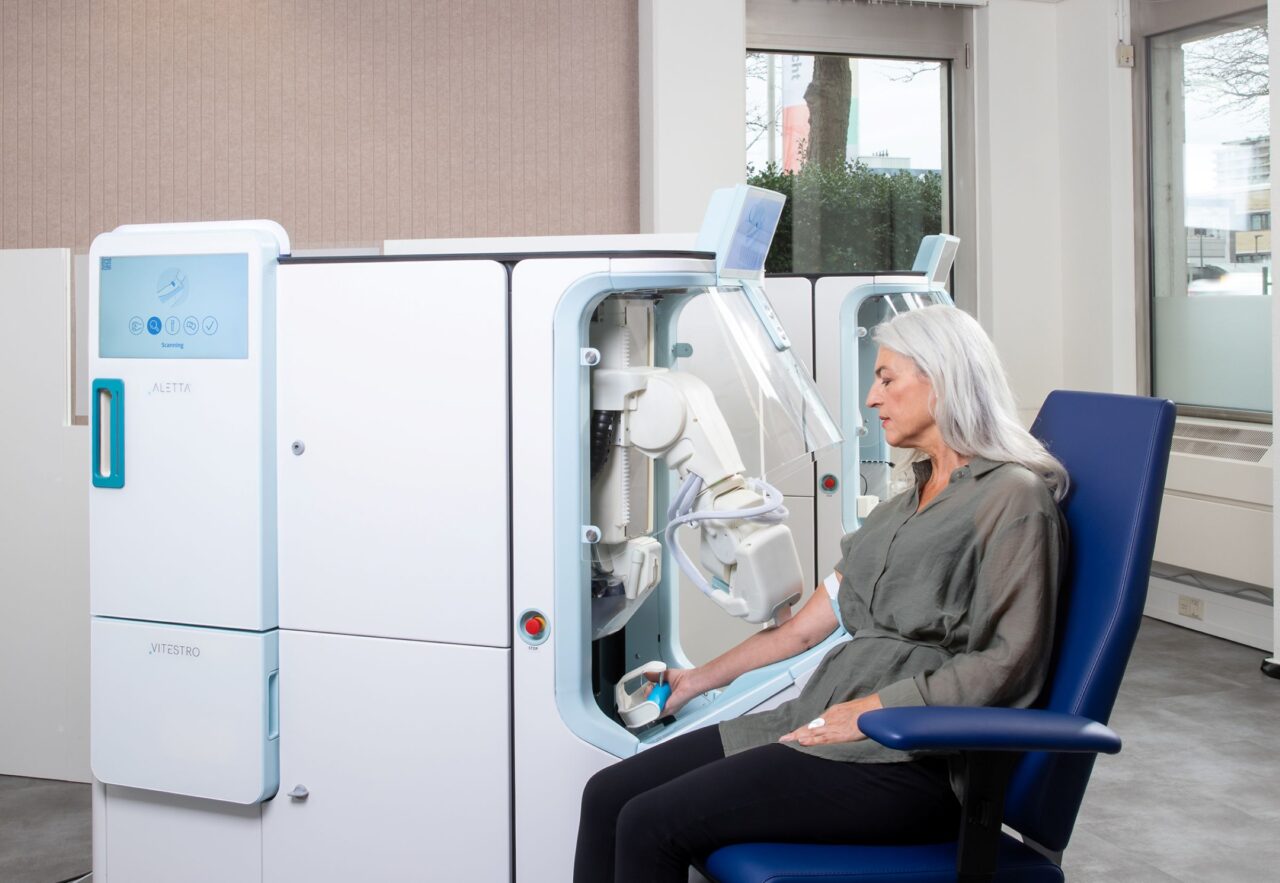
The Aletta system works in a straightforward way. A person places their arm on the device, which then uses near-infrared light, ultrasound and the Doppler imaging technique to map veins in real time. The robotic arm applies a tourniquet, inserts the needle with high precision, collects blood into multiple tubes while inverting them as needed and applies a bandage at the end.
Artificial intelligence controls the entire process to deliver consistent results without the variability that can occur with human phlebotomists/blood draw professionals. One trained supervisor can monitor up to three Aletta units at once, thereby allowing staff to focus on more complex tasks while reducing wait times in busy facilities.
“Vitestro is redefining one of the largest and most under innovated clinical workflows with a first-of-its-kind autonomous robotic platform for diagnostic blood collection addressing an enormous unmet global market need,” said Fred Moll, former CEO of Intuitive Surgical Inc (NASDAQ: ISRG) (ETR: IUI1).
“I believe this technology has the potential to establish a new standard of care, much as robotic surgery did in its early days.”
This funding round builds on Vitestro’s earlier progress. The company previously raised €12 million in a 2023 Series A round to support European market entry followed by an additional US$22 million in 2024 to fund U.S. trial preparations and team expansion.
Vitestro achieved CE marking status in August 2024, which enabled initial deployments at hospitals such as OLVG and Amsterdam UMC in the Netherlands.
Early data from the ongoing Autonomous Blood Drawing Optimization and Performance Testing (ADOPT) trial the company is completing has shown favourable numbers. First-stick success has reached 95 per cent (matching manual draws) and 85 per cent of patients report equal or less pain than with human phlebotomy.
Founders Toon Overbeeke and Brian Joseph started the company in 2017 after facing personal difficulties with blood draws. Overbeeke was frustrated after learning about the struggle his close friend’s father endured as a result of chemotherapy and Joseph complained of waiting excessively for his child’s tests. Chemotherapy can cause traditional manual blood draws to be much more difficult and painful.
Since that time, Vitestro has grown to nearly 100 employees, including engineers, clinicians and commercialization specialists. It currently has an array of open positions to be filled.
Executives aim to make Aletta the standard for routine diagnostic blood collection globally, particularly in high-volume settings where staffing shortages hit hardest.
Switzerland-based Preci-Health is a competitor of note that is also pursuing autonomous blood draw tech and conducting U.S. trials.
Not all robots walk on two legs.
Aletta is a robot that makes drawing blood completely automated.
We live in the future
— Linus ✦ Ekenstam (@LinusEkenstam) January 14, 2026
Read more: Breath Diagnostics advances pre-op pneumonia screening with FDA breakthrough designation
Follow Rowan Dunne on LinkedIn
rowan@mugglehead.com