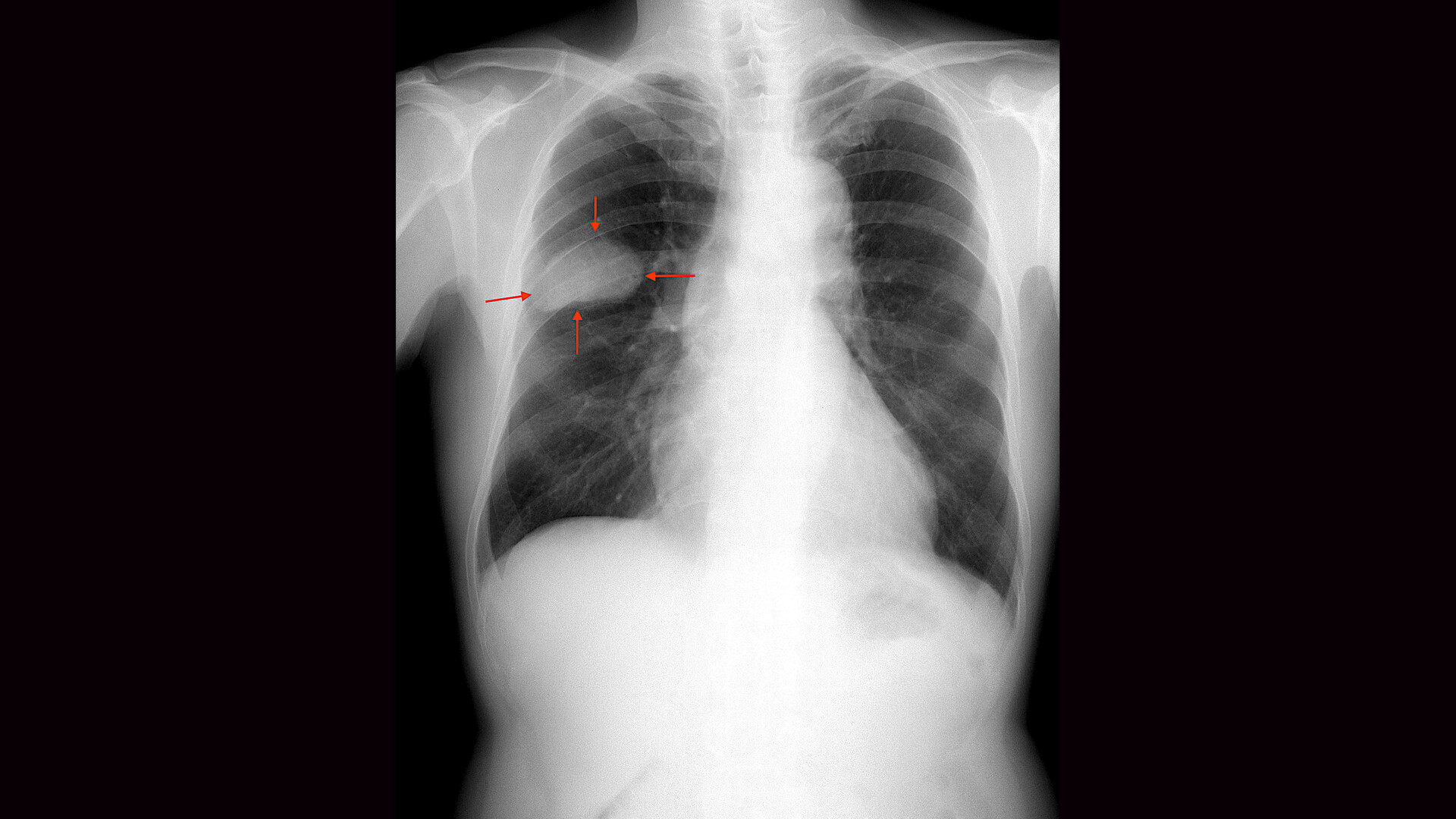
Allarity Therapeutics Inc (NASDAQ: ALLR) has dosed the first patients in a government-backed trial for an aggressive form of lung cancer.
On Feb. 18, doctors at Veterans Affairs hospitals across the United States began giving a new oral drug called stenoparib together with a standard chemotherapy pill (temozolomide) to people whose small cell lung cancer returned after initial treatment. The U.S. Department of Veterans Affairs is fully funding the study and running it at 11 VA medical centres.
Researchers are testing whether this combination shrinks tumours, slows cancer growth and improves survival in patients who have few remaining options. Small cell lung cancer grows quickly and often spreads to the brain. Most patients see their disease come back within months, and only about 40 per cent receive further treatment at that stage.
“When it metastasizes it’s usually game over for these patients within a very short span, three to four months,” said CEO Thomas Jensen in a recent interview.
Stenoparib works by blocking two key processes inside cancer cells: it stops them from repairing damaged DNA and shuts down signals that help tumours resist drugs and grow.
Unlike older similar medicines, it causes fewer severe blood-related side effects, and it reaches the brain, which matters for this type of cancer. Company leaders say early enrolment moves fast because doctors and patients see real need for better choices.
The VA has not issued any statements about this study. The organization funds and sponsors this stenoparib trial for relapsed small cell lung cancer because the disease represents a high-unmet-need area with very limited effective options after initial treatment fails. The VA’s precision oncology initiatives aim to advance biomarker-driven research that could directly improve outcomes for veterans, who often face elevated risks for aggressive lung cancers due to factors like smoking history or service-related exposures.
Skeptical sentiment circulates online
As news of the dosing broke, doubts quickly appeared across investor forums and social media. Some posters label the entire project a “scam,” arguing that similar small biotech announcements often pump stock prices temporarily before reality sets in.
Critics are pointing to the company’s repeated need to sell new shares and question whether positive early talk will translate into actual patient benefits or regulatory approval. They note that Phase 2 trials frequently fail to deliver the hoped-for results, and past company updates have produced mixed market reactions, including sharp drops after earlier trial news.
However, it should be noted that a considerable amount of the critical chatter comes from accounts with questionable credibility.
Read more: Breath Diagnostics advances pre-op pneumonia screening with FDA breakthrough designation
Allarity faces challenges typical of micro-cap biotechs
The micro-cap biotechnology firm has a market value hovering near US$15 million and a stock price floating around 80 to 90 cents.
Allarity has reported steady cash burn from research and has no approved products or revenue. To keep operations running through late 2026 or beyond, it relies on equity financing tools, including a facility that lets it raise up to US$6 million by issuing new shares. This approach risks diluting existing shareholders and putting further downward pressure on the stock.
On the operational front, Allarity is currently running a separate Phase 2 study of stenoparib alone in advanced ovarian cancer. During an interim look last year, the biotech operator reported durable responses/sustained positive reactions in some heavily pretreated patients. This refers to patients with advanced cancer who have already received multiple rounds of different treatments with no success.
Allarity executives have highlighted low toxicity compared with rival drugs and plan further dose testing to meet regulatory demands.
Nonetheless, ongoing losses, a history of reverse stock splits to stay listed on the Nasdaq, and a US$2.5 million SEC civil penalty paid in 2025 over past disclosure issues have made many observers remain wary.
Read more: Prestigious medtech intelligence firm recognizes Breath Diagnostics for innovation
Follow Rowan Dunne on LinkedIn
rowan@mugglehead.com